What is Batten's disease (CLN 6)?
People with Batten disease are born healthy and develop normally during their first years, but over time their development slows, and they gradually lose their intellectual, visual and mobility capacities.
The main types of Battens disease are currently classified according to the genetic defects that affect cells at different levels. There are 14 subsets in total, from CLN1 discovered in 1995 to CLN14 (2021). These different NCL types have symptoms and characteristics in common such as epilepsy, progressive psychomotor impairment and visual deficit. They vary in age of onset and severity, from the newborn stage to adulthood, and this variety is due to different genetic factors, such as heredity and the type of mutation they carry (what is called genotype-phenotype correlation).
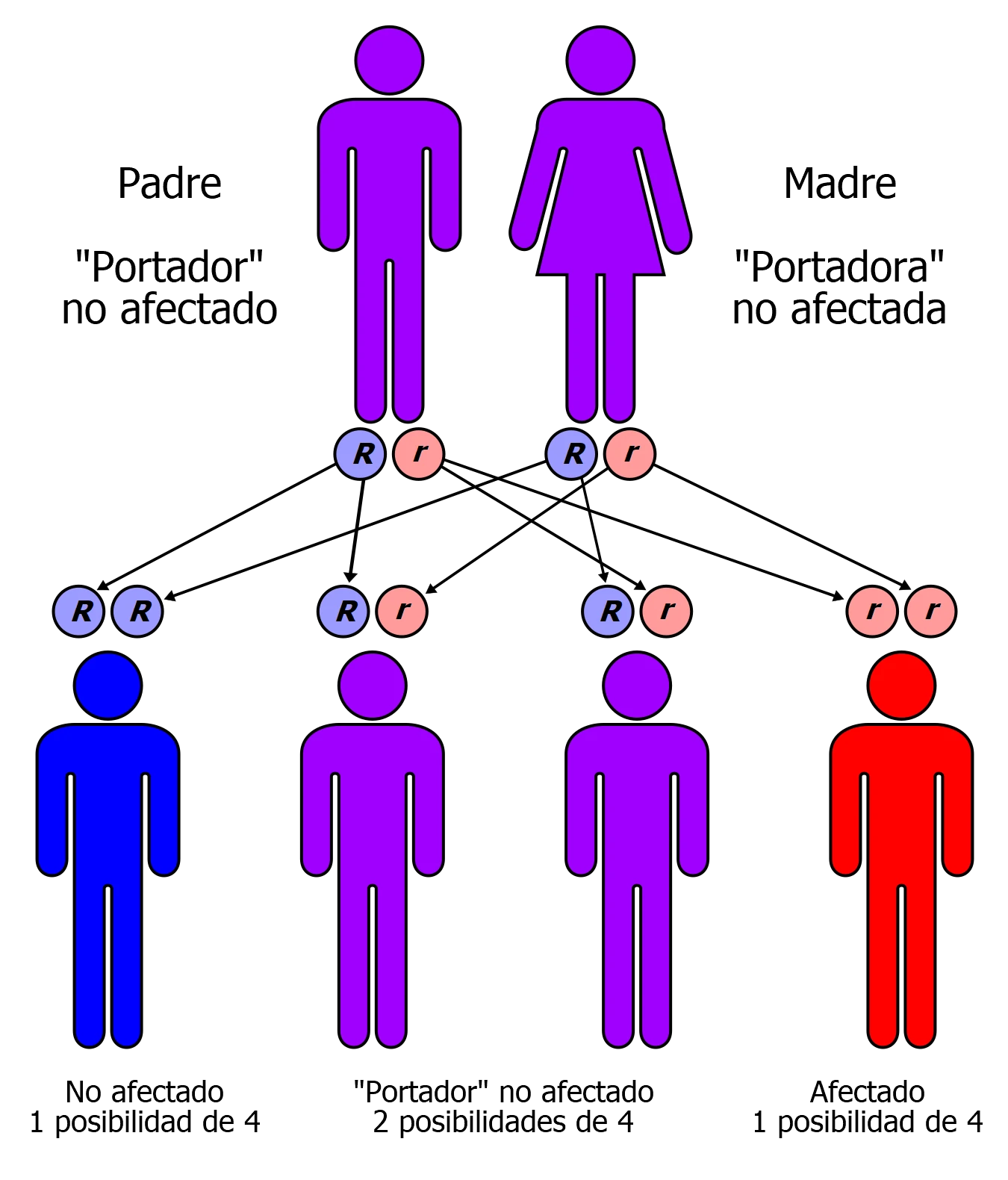
Inheritance is usually autosomal recessive, that is, each parent is a carrier and in each pregnancy of that couple the risk of transmission of the disease is 25%. The gene that is altered is different in each type of NCL and within each gene there may be
very different types of mutations. Thus, in two families with sons or daughters with the same type of NCL (for example, CLN6), they may have different mutations in that same gene (there are more than 70 different ones for CLN6, for example).
From the point of view of age of symptom onset, NCL can be classified into:
– Late infancy NCL appears between the ages of 3-8. In general, these children live to between 8-12 years. This is the group to which Hodei & Ilargi belong: CLN6.
– Juvenile NCL appears between the ages of 5-8. Many of the children survive to adolescence or even to the age of 20.
– Adult NCL appears before the age of 40. These patients can expect to have a normal life.
Symptoms
- Slow intellectual development.
- Changes in personality & behaviour.
- Dementia & confusion.
- Myoclonus-type epileptic seizures, falls, or convulsions.
- Loss of vision.
- Changes in sleep patterns.
- Involuntary muscle spasms.
- Ataxia or instability in walking or standing.
- Loss of ability to speak, walk or communicate.
Ilargi, at the moment has no symptoms but they are expected to start between 4 and 6 years.
This illness is a regressive autosomal disorder. This means that when both parents are carriers of each of the two defective genes that cause the illness, there is a 1 in 4 (25%) chance of transmission to any of their children.
Without appropriate treatment, this illness causes rapid neurological decline, resulting in a rapid decline of abilities to walk, talk, think & see.
To date, the only treatment that exists, with the potential to be a disease course modifier, is a gene therapy developed by Amicus Therapeutics that is in the clinical trial phase, which has been conducted at Nationwide Children’s Hospital in Columbus, Ohio.
This treatment consists of replacing the mutated gene with one which is transported to the brain by a harmless virus.
Useful information, studies and publications on Batten disease
- Instituto Nacional de Trastornos Neurológicos y Accidentes Cerebrovasculares. Definicion, tratamientos, pronosticos y estudios clinicos de la enfermedad BATTEN
- Amicus Therapeutics. Amicus anuncia datos clínicos provisionales positivos adicionales para la terapia génica de la enfermedad de Batten CLN6…(inglés)
- Elsevier Inc. La terapia génica corrige patologías cerebrales y conductuales en la enfermedad de CLN6-Batten.. (inglés)
- Batten Disease. Viviendo con la enfermedad de Batten (inglés)
- Joana Carvalho. Una sustáncia química podría ayudar a los afectados por la enfermedad de Batten a prevenir la muerte celular. (inglés)
- Batten Disease News. Scientist Wins Grant to Study CLN6 Batten, Advance Early Trials – Batten Disease News
- Europapress. Investigador de la USAL avanza en identificación de nuevos mecanismos neurodegenerativos en la enfermedad de Batten (europapress.es)
Revised & recommended scientific references
- Lopez-Fabuel I, Garcia-Macia M, Buondelmonte C, Burmistrova O, Bonora N, Alonso-Batan P, Morant-Ferrando B, Vicente-Gutierrez C, Jimenez-Blasco D, Quintana-Cabrera R, Fernandez E, Llop J, Ramos-Cabrer P, Sharaireh A, Guevara-Ferrer M, Fitzpatrick L, Thompton CD, McKay TR, Storch S, Medina DL, Mole SE, Fedichev PO, Almeida A, Bolaños JP. Aberrant upregulation of the glycolytic enzyme PFKFB3 in CLN7 neuronal ceroid lipofuscinosis. Nat Commun. 2022 Jan 27;13(1):536. doi: 10.1038/s41467-022-28191-1. PMID: 35087090
In this original article, the main researchers, from the University of Salamanca, have found in a CLN7 mouse model, a failure in autophagy (waste cleaning system in cells) that produces an accumulation of altered mitochondria. This produces an increase in free radicals in the mitochondria that is mediated by an enzyme, PFKFB3, which is activated when it shouldn’t. These researchers apply a PFKFB3 blocker called AZ67 to the affected cells and they improve, disappearing the stress situation in the CLN7 cells. They conclude that designing therapies targeting the PFKFB3 enzyme could be a new therapeutic line in lysosomal diseases.
- Specchio N, Ferretti A, Trivisano M, Pietrafusa N, Pepi C, Calabrese C, Livadiotti S, Simonetti A, Rossi P, Curatolo P, Vigevano F. Neuronal Ceroid Lipofuscinosis: Potential for Targeted Therapy. Drugs. 2021 Jan;81(1):101-123. doi: 10.1007/s40265-020-01440-7. PMID: 33242182.
In this review article, the authors of the Bambino Gesú Hospital in Rome, specialists in epilepsy and neurogenetic diseases, review the therapeutic lines currently available for each type of NCL.
- Mole SE, Cotman SL. Genetics of the neuronal ceroid lipofuscinoses (Batten disease). Biochim Biophys Acta. 2015 Oct;1852(10 Pt B):2237-41. doi: 10.1016/j.bbadis.2015.05.011. PMID: 26026925.
In this review article, the authors, from University College London, update on the genetics of NCL and possible genotype-phenotype correlations. That is, if there is an association between a specific mutation and the earlier or later onset of symptoms, or that these symptoms are milder or more severe.